 
In 1980, the CIPM clarified the above definition, defining that the carbon-12 atoms are unbound and in their ground state. In 1961, the isotope carbon-12 was selected to replace oxygen as the standard relative to which the atomic weights of all the other elements are measured. 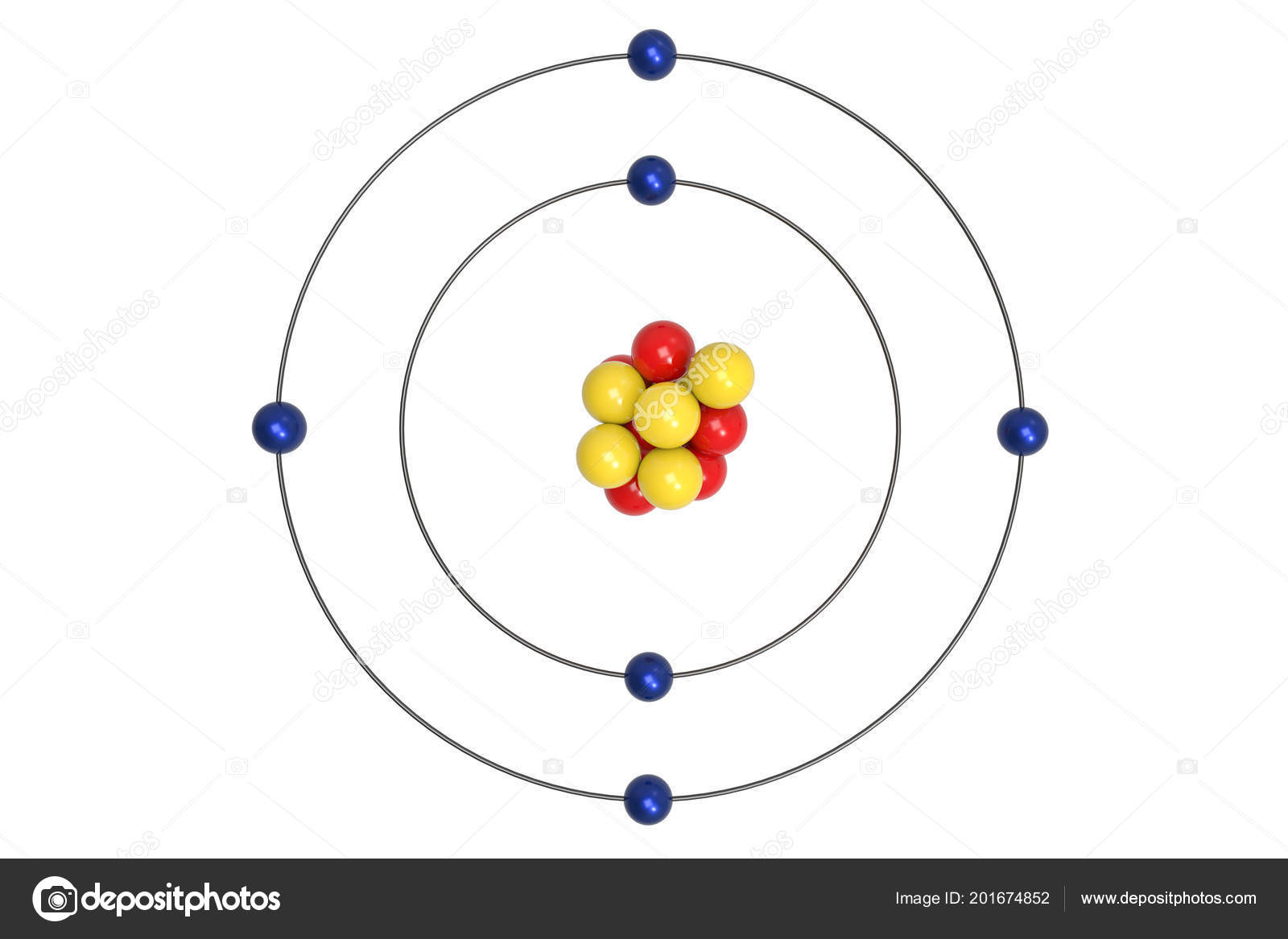
This was adopted by the CIPM (International Committee for Weights and Measures) in 1967, and in 1971, it was adopted by the 14th CGPM (General Conference on Weights and Measures). Mole is the amount of substance of a system which contains as many elementary entities as there are atoms in 12 gram of carbon 12 its symbol is "mol". The two organizations agreed in 1959/60 to define the mole as follows. Carbon-12 is composed of 6 protons, 6 neutrons, and 6 electrons.īefore 1959, both the IUPAP and IUPAC used oxygen to define the mole the chemists defining the mole as the number of atoms of oxygen which had mass 16 g, the physicists using a similar definition but with the oxygen-16 isotope only. Carbon-12 is of particular importance in its use as the standard from which atomic masses of all nuclides are measured, thus, its atomic mass is exactly 12 daltons by definition. Carbon-12, 12C GeneralĬarbon-12 ( 12C) is the most abundant of the two stable isotopes of carbon ( carbon-13 being the other), amounting to 98.93% of element carbon on Earth its abundance is due to the triple-alpha process by which it is created in stars. For the building in Oregon, see Carbon12. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
